
A key feature of suspensions is that the suspended particles settle over time if left undisturbed.Ĭolloids are heterogeneous like suspensions but visually appear to be homogeneous because the particles in the mixture are very small-1 nanometer to 1 micrometer. 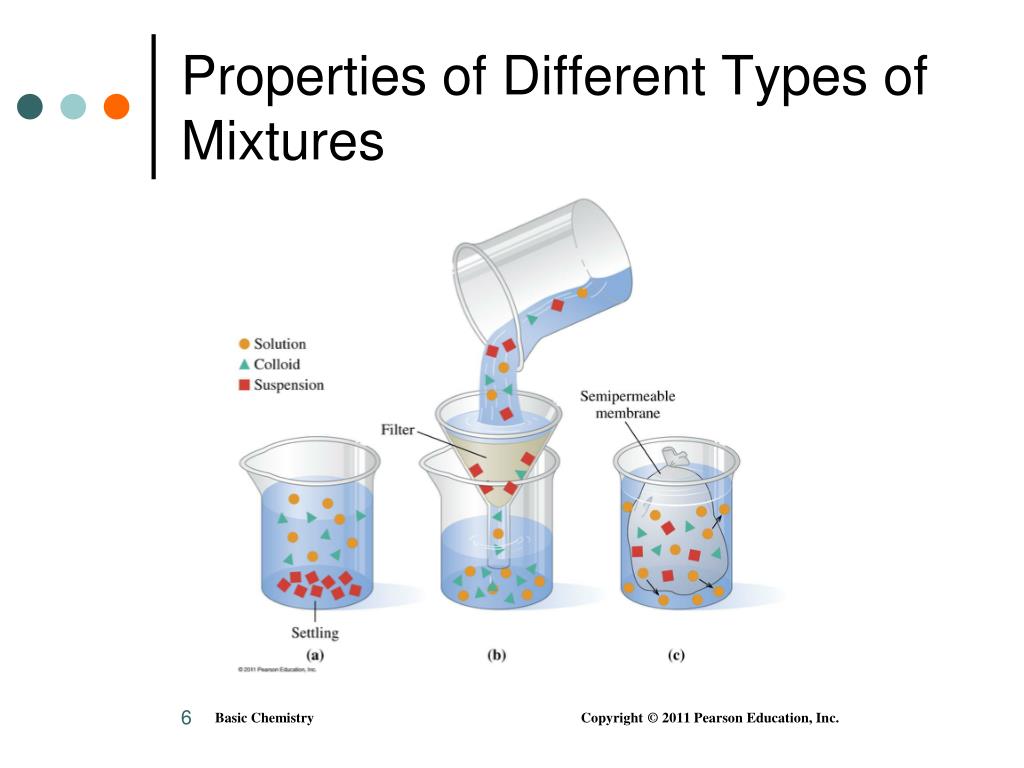
They are bigger than 1 micrometer and are usually large enough to be visible to the naked eye. The solid particles do not dissolve in the solvent but are suspended and freely floating. Solid in solid: Alloys like steel, brass or bronze are an example of such mixtures.Ī suspension is a heterogeneous mixture that contains solid particles that are large enough for sedimentation.Liquid in solid: Examples of this include mercury in gold, forming an amalgam, and water (moisture) in salt.Gas in solid: An example of this is hydrogen dissolved in palladium.Solid solutions: Solid solvents can also dissolve solutes of any state of matter.Many solid in liquid mixtures are not homogeneous so they aren't solutions. Solid in liquid: Sugar or salt solutions in water are examples of such mixtures.Liquid in liquid: Example include alcoholic beverages they are solutions of ethanol in water.Gas in liquid: Examples include oxygen in water, or carbon dioxide in water.Liquid solutions: Liquid solvents are capable of dissolving any type of solutes.The most common example of a gaseous solution is the air in our atmosphere, which is nitrogen (the solvent) and has solutes like oxygen and other gases. Gaseous solutions: When the solvent is a gas, it is only possible to dissolve gaseous solutes in it.The solute assumes the phase (solid, liquid or gaseous) of the solvent when the solvent is the larger fraction of the mixture. Not only that, the individual components of the solution can be different states of matter. Solutions can be liquid, gaseous or even solid. The ratio of mass of the solute to the solvent is called the concentration of the solution. When the solvent is water, it is called an aqueous solution. Solutions are homogeneous mixtures that contain a solute dissolved in a solvent, e.g. Solutions are homogeneous while suspensions and colloids are heterogeneous. There are three families of mixtures: solutions, suspensions and colloids. Many substances, such as salt and sugar, dissolve in water to form homogeneous mixtures. Even the air that's in our atmosphere is a homogeneous mixture of various gases and-depending upon the city you live in-pollutants. Even a mixture of oil and water is heterogeneous because the density of water and oil is different, which prevents uniform distribution in the mixture.Įxamples of homogeneous mixtures are milkshakes, blended vegetable juice, sugar dissolved in coffee, alcohol in water, and alloys like steel. Examples of Homogeneous and Heterogeneous MixturesĮxamples of heterogeneous mixtures would be ice cubes (before they melt) in soda, cereal in milk, various toppings on a pizza, toppings in frozen yogurt, a box of assorted nuts. They can often be visually told apart and even separated relatively easily, although many methods exist to separate homogeneous solutions as well.Ī visualization for the differences between substances (compounds, elements) and mixtures (both homogenous and heterogenous). This uniformity is because the constituents of a homogeneous mixture occur in the same proportion in every part of the mixture.Ĭonversely a heterogeneous mixture is one where the constituent substances are not uniformly distributed. Their composition is uniform i.e., same throughout the mixture. Commonly called solutions, homogeneous mixtures are those where the substances mix so well that they cannot be individually seen in a differentiated, distinct form. But in a mixture, elements and compounds are both found intermingled physically but not chemically-no atomic bonds form between the pure substances that constitute the mixture.īut regardless of atomic bonds, mixtures can become quite cohesive. Molecules are made of atoms that have bonded together. Compounds are pure substances because they only contain one type of molecule. The difference between a mixture and a compound is how the elements or substances combine to form them. All mixtures comprise two or more pure substances (elements or compounds).
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
